Abstract
This paper explores the neural correlates of posture and balance after stroke, and provides visual examples of postural instability in a patient with watershed and midbrain stroke. A full stroke physiatry workup of the patient and the case report could be found at the beginning of the video (Link at the bottom).
- 1- Introduction
- 2- Neuroanatomy of posture: the descending pathway
- 3- Changes in posture during walking
- 4- Postural Orientation Is Important for Optimizing Execution of Tasks, Interpreting Sensations, and Anticipating Disturbances to Balance
- 5- Patient’s video (Neurorehabilitation and truncal instability)
- 6- References
*Note: Prior to publication of any patient-related medical information and images, a written consent form is obtained and data are anonymized. We are eternally grateful to these amazing patients for allowing us to publish their anonymized information and images. We appreciate their contribution to stroke research and training.
1- Introduction
The control of posture is an important element in almost any motor task. The two components of posture, orientation and balance, require continuous adjustment, and involve several sensory systems (Kandel et al., 2013).
To appreciate the complexity of maintaining balance and orientation, imagine that you are carrying a tray of glasses full of water on a boat. You have a tray full of drinks to be delivered to a table on the other side of the rolling deck. Even as your mind is occupied with other thoughts-such as how much you dislike some of the guests and wished you could taint their drink, unconscious processes allow you to maintain your balance and walk as the boat moves on the water. The apparently simple task of delivering drinks is supported by a truly complex sensorimotor process for controlling postural orientation and balance. As you cross the deck your brain rapidly processes sensory information and adjusts motor output to maintain your balance, the upright orientation of your head and trunk, and stable arms supporting the tray of full glasses. Before you reach out to place a glass on the table, your nervous system makes anticipatory postural adjustments to maintain your balance. Sudden unexpected motions of the boat evoke automatic postural responses that prevent falls. Somatosensory, vestibular, and visual information is integrated to provide a coherent picture of the position and velocity of the body in space and to generate and update motor commands that maintain balance and orientation.
Postural equilibrium, or balance, involves active resistance to external forces acting on the body. The dominant external force affecting equilibrium on earth is gravity. Postural orientation is the positioning of body segments with respect to each other and to the environment. Depending on the particular task or behaviour, body segments may be aligned with respect to gravitational vertical, visual vertical, or the support surface. The biomechanical requirements of postural control depend on anatomy and postural orientation and thus vary with the animal. Nevertheless, in a variety of species the control mechanisms for postural equilibrium and orientation have many common features. The sensorimotor mechanisms for postural control are quite similar in humans and quadrupedal mammals even though their habitual stance is different Kandel et al., 2013; (Ivanenko & Gurfinkel, 2018). With many segments linked by joints, the body is mechanically unstable. To maintain balance the nervous system must control the position and motion of the body’s centre of mass as well as the body’s rotation about its centre of mass. The centre of mass is a point that represents the average position of the body’s total mass. In the standing cat, for example, the centre of mass is located in the trunk just rostral to the midpoint between forelimbs and hind limbs (Kandel et al., 2013).
Although gravity pulls on all body segments, the net effect on the body acts through the centre of mass. The force of gravity is opposed by the ground reaction force, which pushes upward against each foot (Forghany et al., 2011). The net ground reaction force occurs at an imaginary point on the ground called the centre of pressure (Verheyden et al., 2014). The location of the centre of mass in the body is not fixed but depends on postural orientation. When you are standing upright, for example, your centre of mass is located in the abdomen approximately 20 mm in front of the second lumbar vertebra. When you flex at the hips, however, the centre of mass moves forward to a position outside the body. Maintaining balance while standing requires keeping the downward projection of the centre of mass within the base of support, an imaginary area defined by those parts of the body in contact with the environment (Kandel et al., 2013; Forghany et al., 2011).
Upright stance requires two actions:
(1) maintaining support against gravity (keeping the center of mass at some height)
(2) maintaining balance (controlling the trajectory of the centre of mass in the horizontal plane). Balance and antigravity support are controlled separately by the nervous system and may be differentially affected in certain pathological conditions.
Antigravity support, or postural tone, represents the tonic activation of muscles that generate force against the ground to keep the limbs extended and the centre of mass at the appropriate height. In humans much of the support against gravity is provided by passive bone-on-bone forces in joints such as the knees, which are fully extended during stance, and in stretched ligaments such as those at the front of the hips. Nevertheless, antigravity support in humans also requires active muscle contraction, for example in ankle, trunk, and neck extensors. Tonic activation of antigravity muscles is not sufficient, however, for maintaining balance (Kandel et al., 2013).
Both bipeds and quadrupeds are inherently unstable, and their bodies sway during quiet stance. Actively contracting muscles exhibit a spring-like stiffness that helps to resist body sway, but muscle stiffness alone is insufficient for maintaining balance. Likewise, stiffen- ing of the limbs through muscle co-contraction is not sufficient for balance control. Instead, complex pat- terns of muscle activation produce direction-specific forces to control the body’s center of mass. Body sway caused by even subtle movements, such as the motion of the chest during breathing, is actively counteracted by the posture control system (Takakusaki, 2017).
When a sudden disturbance causes the body to sway, various motor strategies are used to maintain the centre of mass within the base of support. In one strategy the base of support remains fixed relative to the support surface. While the feet remain in place the body rotates about the ankles back to the upright position. In other strategies the base of support is moved or enlarged, for example by taking a step or by grabbing a support with the hand (Takakusaki, 2017).
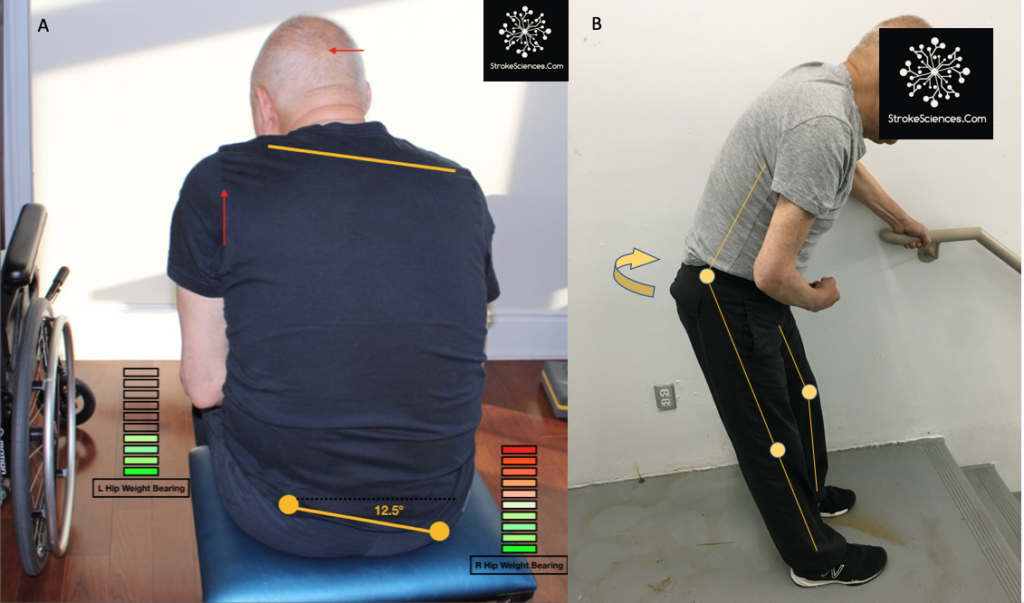
Older views of motor control focused on trunk and proximal limb muscles as the main postural effectors. Recent behavioural studies show that any group of muscles from the neck and trunk, legs and arms, or feet and hands can act as postural muscles depending on the body parts in contact with the environment and the biomechanical requirements of equilibrium (as shown in figure1)
2- Neuroanatomy of posture: the descending pathway
A little neuroanatomy might help visualize the neural systems involved in posture. The cerebellum influences posture and movement through its connections in the brain stem motor nuclei, which can directly modulate spinal motor circuits (Kandel et al., 2013). However, the major influence of the cerebellum on movement is through its connections in the ventrolateral nuclei of the thalamus, which connect directly to the motor and premotor cortex.
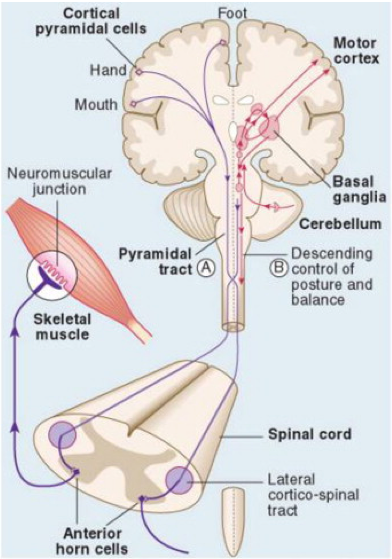
The basal ganglia are a collection of subcortical nuclei (see Figure 16–9) that receive direct projections from much of the neocortex, including sensory, motor, and premotor areas, and those parts of association cortex that are important for motivation, cognition, and emotion. The output nuclei of the basal ganglia send signals to regions of the thalamus that project to the cerebral cortex. Although the functions of the basal gan- glia have remained surprisingly elusive, dysfunction of the basal ganglia results in particularly striking disorders of movement characteristic of Parkinson disease (tremor at rest, rigidity, and disinclination to move) and of Huntington disease (choreiform movements) (Harley et al., 2006; Ivanenko & Gurfinkel, 2018; Verheyden et al., 2014).
Thus an important consequence of basal ganglia dysfunction is that the abnormal signals sent to cortical motor areas have a major, negative, impact on motor output. Indeed, cortical lesions that limit voluntary movement also abolish the involuntary movements associated with disorders of the basal ganglia. This capacity of the basal ganglia to create marked disorders of movement when they are functioning abnormally must in some way be matched by a similar profound influence over normal motor function.
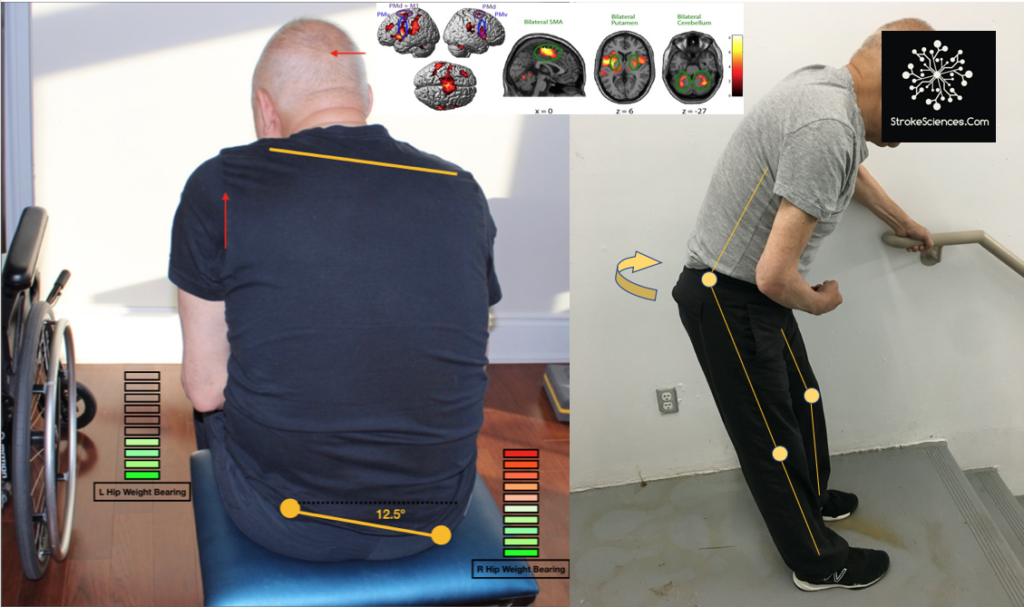
Moving down the descending pathway, we see that the motor information carried in the corticospinal tract is significantly modulated by a continuous stream of information from other motor regions as well as tactile, visual, and proprioceptive information needed to make voluntary movement both accurate and properly sequenced (Harley et al., 2006). As an example in figure 3, we see a patient with internal capsule and midbrain stroke (cerebral peduncle infarct) unable to maintain upright posture in sitting. Due to prior falls and diminished proprioceptive input, this patient continuously feels as if he is falling forward and to the side of the hemiparesis ( right side). strategies differ based on the support provided to ensure the patient is not falling. In the case of sitting, the backrest of the wheelchair provides the support and peace of mind for the patient, the moment patient is transferred to an unsupported stool without a backrest, patient overcorrects to feel the wall behind him. In standing, aside from postural compensatory mechanisms, such as truncal rotation, knee over extension, and foot flattening, the patient also leans heavily toward the parson assisting him. Note that patient’s head is heavily acting as a balance mechanism to move his weight away from the affected side (hemiparetic side)
Also, the medial brain stem system originates in portions of the reticular formation, vestibular nuclei, and superior colliculus. This system receives information from the cortex and other motor centres for the control of posture and locomotion. The lateral brain stem system originates from the red nucleus. It receives input from the cortex as well but is involved in the control of arm and hand movements. It is important to note that spinal motor circuits are not regulated solely by descending commands. Reflex circuits and pattern generators within the spinal cord can coordinate stereotyped movements such as stepping without descending signals. A newborn infant, whose descending pathways cannot yet control the spinal cord, is able to execute stepping movements when lifted into the air. Descending systems coordinate reflex and patterned movements generated by spinal motor circuits and can even create new patterns of muscle activation through direct action on motor neurons. This cortical control enables greater flexibility (Takakusaki et al., 2016).
3- Changes in posture during walking
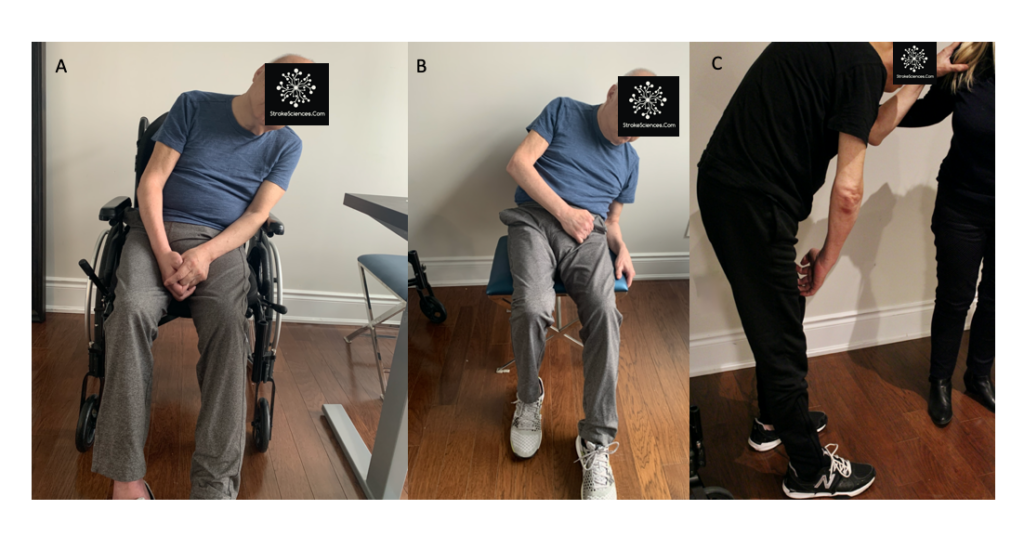
A common preparatory postural adjustment occurs when one begins to walk. The centre of mass is accelerated forward and laterally by the unweighting of one leg (Kandel et al., 2013). This postural adjustment appears to be independent of the stepping program that underlies ongoing locomotion. Similarly, a forward shift of the centre of mass precedes the act of standing on the toes. A subject is unable to remain standing on his toes if he simply activates the calf muscles without moving his centre of mass forward; he rises onto his toes only momentarily before gravity restores a flat-footed stance. Moving the centre of mass forward over the toes before activating the calf muscles aligns it over the anticipated base of support and thus stabilizes the toe stance. In figure 1-B, you see the patient elaborate compensatory changes to the hips, knees and ankles, as well causing a flexed posture and a flat position of right foot (Takakusaki et al., 2016).
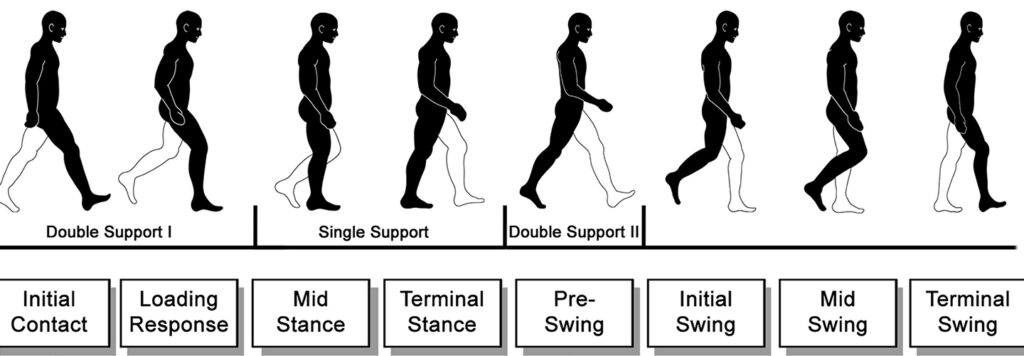
Locomotion, too, has an important postural component. During walking and running the body is in a constant state of falling as the centre of mass moves forward and laterally toward the leg that is in the swing phase (Forghany et al., 2011; Harley et al., 2006). The centre of mass is within the base of support during walking only when both feet are on the ground, the double stance phase, and not at all during running. When one foot is supporting the body, the centre of mass moves forward in front of the foot, always medial to the base of support. Falling is prevented during walking and running by moving the base of support forward and laterally under the falling centre of mass. Postural equilibrium during gait relies on the appropriate placement of each step to control the speed and trajectory of the centre of mass. The nervous system plans foot placement several steps in advance using visual information about the terrain and surrounding environment (Forghany et al., 2011; Harley et al., 2006).
4- Postural Orientation Is Important for Optimizing Execution of Tasks, Interpreting Sensations, and Anticipating Disturbances to Balance
Despite the fact, that postural orientation interacts with balance control, the two systems can act independently. The energy needed to maintain body position over a period of time can influence postural orientation. In humans, for example, the upright orientation of the trunk with respect to gravity minimizes the forces and thus the energy required to hold the body’s centre of mass over the base of support.this is evident in figure 2, since the centra nervous system develops and tunes based on intact sensory and motor mechanisms, after stroke the brain still works based on its “energy conservation” rule (Harley et al., 2006; Ivanenko & Gurfinkel, 2018; Takakusaki, 2017). Though, in the absence of optimal motor performance -due to stroke- peculiar compensatory mechanisms emerge.
5. Patient’s video (Neurorehabilitation and truncal instability)
In this video, a complete stroke physiatry work up is presented for a patient with postural instability.
6- References
14.3 Motor Responses – Anatomy and Physiology. (n.d.). Retrieved May 16, 2020, from https://opentextbc.ca/anatomyandphysiology/chapter/14-3-motor-responses/
Effects of continuous visual feedback during sitting balance training in chronic stroke survivors. Retrieved May 16, 2020, from https://www.ncbi.nlm.nih.gov/pmc/articles/PMC5644142/
Forghany, S., Tyson, S., Nester, C., Preece, S., & Jones, R. (2011). Foot posture after stroke: Frequency, nature and clinical significance. Clinical Rehabilitation, 25(11), 1050–1055. https://doi.org/10.1177/0269215511410581
Functions and dysfunctions of the basal ganglia in humans. (n.d.). Retrieved May 16, 2020, from https://www.jstage.jst.go.jp/article/pjab/94/7/94_PJA9407B-02/_html/-char/en
Harley, C., Boyd, J. E., Cockburn, J., Collin, C., Haggard, P., Wann, J. P., & Wade, D. T. (2006). Disruption of sitting balance after stroke: Influence of spoken output. Journal of Neurology, Neurosurgery, and Psychiatry, 77(5), 674–676. https://doi.org/10.1136/jnnp.2005.074138
Kandel, E. R., Schwartz, J., Jessel, T., Sieglebaum, S., & Hudspeth, A. J. (n.d.). Principles of Neural Science, Fifth Edition | AccessNeurology | McGraw-Hill Medical. Retrieved May 10, 2020, from https://neurology.mhmedical.com/book.aspx?bookID=1049
Ivanenko, Y., & Gurfinkel, V. S. (2018). Human Postural Control. Frontiers in Neuroscience, 12. https://doi.org/10.3389/fnins.2018.00171
Takakusaki, K. (2017). Functional Neuroanatomy for Posture and Gait Control. Journal of Movement Disorders, 10(1), 1–17. https://doi.org/10.14802/jmd.16062
Takakusaki, K., Chiba, R., Nozu, T., & Okumura, T. (2016). Brainstem control of locomotion and muscle tone with special reference to the role of the mesopontine tegmentum and medullary reticulospinal systems. Journal of Neural Transmission, 123(7), 695–729. https://doi.org/10.1007/s00702-015-1475-4
Verheyden, G., Ruesen, C., Gorissen, M., Brumby, V., Moran, R., Burnett, M., & Ashburn, A. (2014). Postural alignment is altered in people with chronic stroke and related to motor and functional performance. Journal of Neurologic Physical Therapy: JNPT, 38(4), 239–245. https://doi.org/10.1097/NPT.0000000000000054